Contents
Description
Calcium is an element of the main subgroup II of group IV of the periodic system of chemical elements of D.I. Mendeleev, has an atomic number of 20 and an atomic mass of 40.08. The accepted designation is Ca (from Latin – Calcium).
Calcium history
Calcium was discovered in 1808 by Humphrey Davy, who, by electrolysis of slaked lime and mercury oxide, obtained a calcium amalgam, as a result of the process of distilling mercury from which a metal, called calcium, remained. In Latin, lime sounds like calx, and it was this name that was chosen by the English chemist for the open substance.
Physical and chemical properties

Calcium is a reactive, soft, silvery-white alkali metal. Due to the interaction with oxygen and carbon dioxide, the metal surface grows dull, therefore calcium needs a special storage mode – a tightly closed container in which the metal is poured with a layer of liquid paraffin or kerosene is mandatory.
Daily requirement for calcium
Calcium is the most famous of the trace elements necessary for a person, the daily requirement for it is from 700 to 1500 mg for a healthy adult, but it increases during pregnancy and lactation, this must be taken into account and calcium should be obtained in the form of preparations.
In nature
Calcium has a very high chemical activity, therefore it does not occur in nature in its free (pure) form. Nevertheless, it is the fifth most common in the earth’s crust, in the form of compounds it is found in sedimentary (limestone, chalk) and rocks (granite), anorite feldspar contains a lot of calcium.
In living organisms it is widespread enough, its presence is found in plants, animals and humans, where it is present mainly in the composition of teeth and bone tissue.
Calcium-rich foods

Sources of calcium: dairy and dairy products (the main source of calcium), broccoli, cabbage, spinach, turnip leaves, cauliflower, asparagus. Calcium also contains egg yolks, beans, lentils, nuts, figs (calorizator). Another good source of dietary calcium is the soft bones of salmon and sardines, any seafood. The champion in calcium content is sesame, but only fresh.
Calcium must enter the body in a certain ratio with phosphorus. The optimal ratio of these elements is considered to be 1: 1.5 (Ca: P). Therefore, it is correct to eat foods rich in these minerals at the same time, for example, beef liver and liver of fatty fish, green peas, apples and radishes.
Calcium absorption
An obstacle to the normal absorption of calcium from food is the consumption of carbohydrates in the form of sweets and alkalis, which neutralize the hydrochloric acid in the stomach, which is necessary for the dissolution of calcium. The process of assimilating calcium is quite complicated, so sometimes it is not enough to get it only with food, an additional intake of a trace element is required.
Interacting with others
To improve the absorption of calcium in the intestine, vitamin D is needed, which tends to facilitate the absorption of calcium. When taking calcium (in the form of supplements) in the process of eating, the absorption of iron is blocked, but taking calcium supplements separately from food does not affect this process in any way.
Useful properties of calcium and its effect on the body
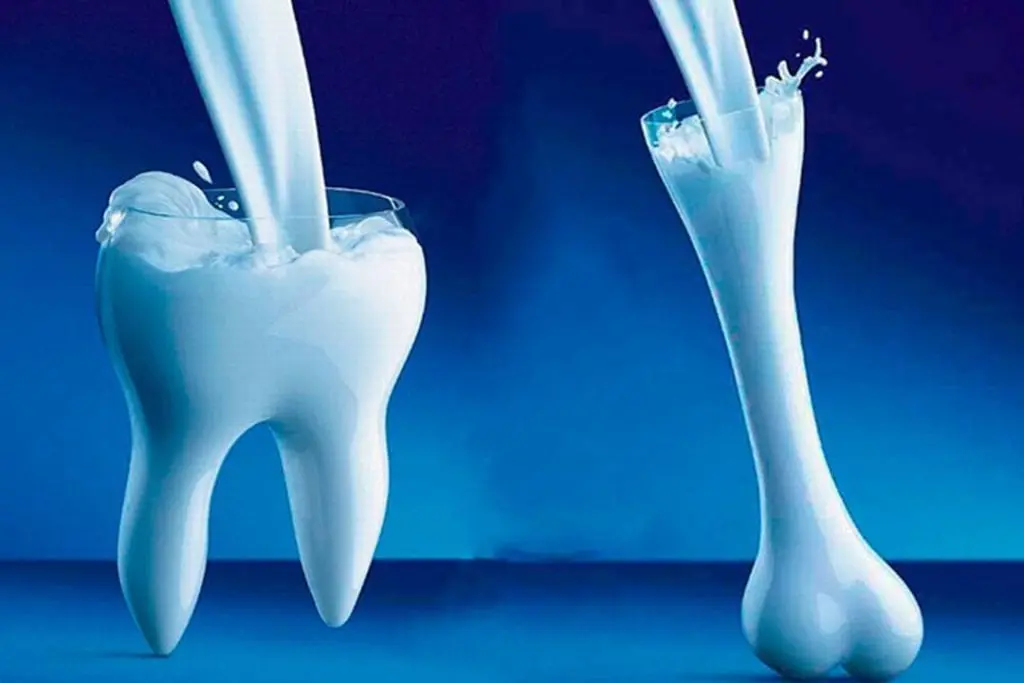
Almost all of the body’s calcium (from 1 to 1.5 kg) is found in bones and teeth. Calcium is involved in the processes of excitability of nervous tissue, muscle contractility, blood coagulation processes, is a part of the nucleus and membranes of cells, cellular and tissue fluids, has antiallergic and anti-inflammatory effects, prevents acidosis, activates a number of enzymes and hormones. Calcium is also involved in the regulation of the permeability of cell membranes, has an effect opposite to sodium.
Signs of calcium deficiency
Signs of a lack of calcium in the body are the following, at first glance, unrelated symptoms:
- nervousness, deterioration of mood;
- cardiopalmus;
- cramps, numbness of the limbs;
- retarded growth and children;
- high blood pressure;
- delamination and fragility of nails;
- joint pain, lowering the “pain threshold”;
- profuse menstruation.
- Causes of calcium deficiency

The causes of calcium deficiency can be unbalanced diets (especially fasting), low calcium in food, smoking and craving for coffee and caffeine-containing drinks, dysbiosis, kidney disease, thyroid gland, pregnancy, lactation and menopause.
Signs of calcium excess
Excess calcium, which can occur with excessive consumption of dairy products or uncontrolled use of medications, is characterized by intense thirst, nausea, vomiting, loss of appetite, weakness, and increased urination.
The use of calcium in usual life
Calcium has found application in the metallothermic production of uranium, in the form of natural compounds it is used as a raw material for the production of gypsum and cement, as a disinfectant (well-known bleach).










